Presented at CSHL
annual structural biology meeting on Aug 8, 2001. Cold Spring Harbor, NY.
Protein family analysis by
Valentin A.ILYIN and Andrej SALI
The Rockefeller
University
The Rockefeller
University
Integration
of multiple structures view, sequence alignments and homology tree clustering
analysis in one application is important for understanding biological problems
in proteins and protein families. Recently an attempt was made to create such
application http://salilab.org/modview
(Ilyin et. al, 2001).
Comparative
analysis of multiple proteins is essential part in sequence-structure function
relationship, mapping the protein function in the metabolism pathways,
recognition of similarities and differences between different organisms.
Here
we present an analysis of two protein families of DNA binding proteins by
ModView. Zinc
finger protein (PDB code 1mey, Kim and Berg, 1996) and The DNA-Binding Domain Of
The Replication Initiation Protein E1 From Papillomavirus (PDB code 1f08,
Enemark et. Al, 2000).
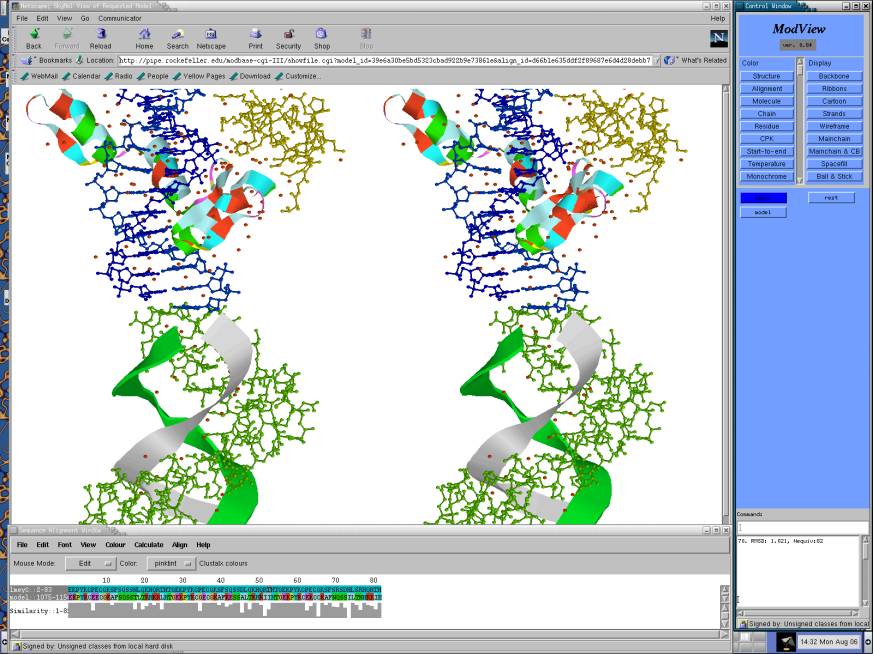
Fig 1 General picture of ModView (Ilyin et. al, 2001). There are two main windows multiple structure viewer and sequence alignment editor and also the control window. ModView as Netscape plug-in presents structures and sequences located at local or remote resources.
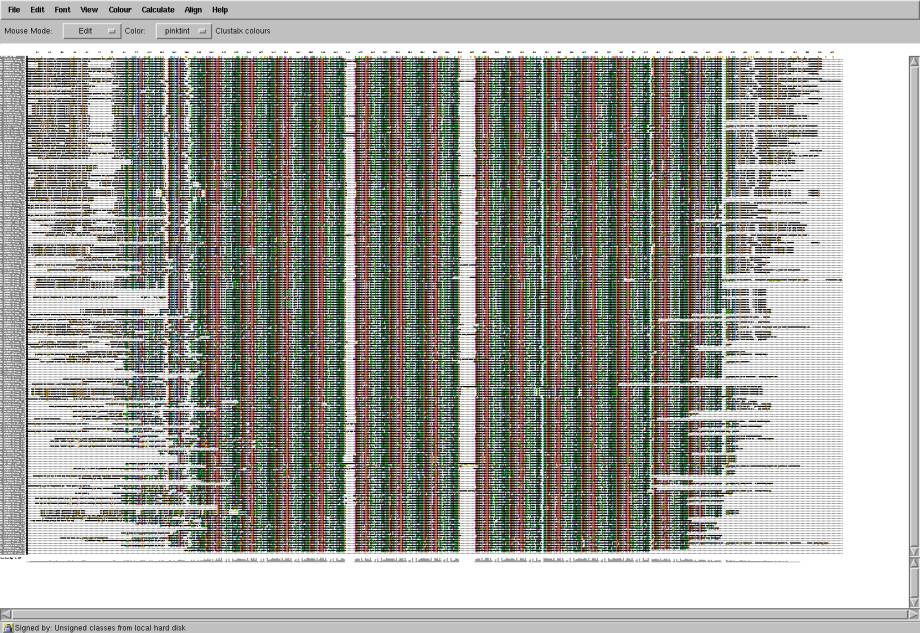
Fig 2. The alignment of zinc finger DNA-binding protein super family. All residues are colored by residue type. Conservative residues have more dense color. Repetitions of the same structural motif in each particular sequence and across the whole super family is clearly seen from the alignment. This motif binds with DNA. There are also variations in N and C terminals of proteins.
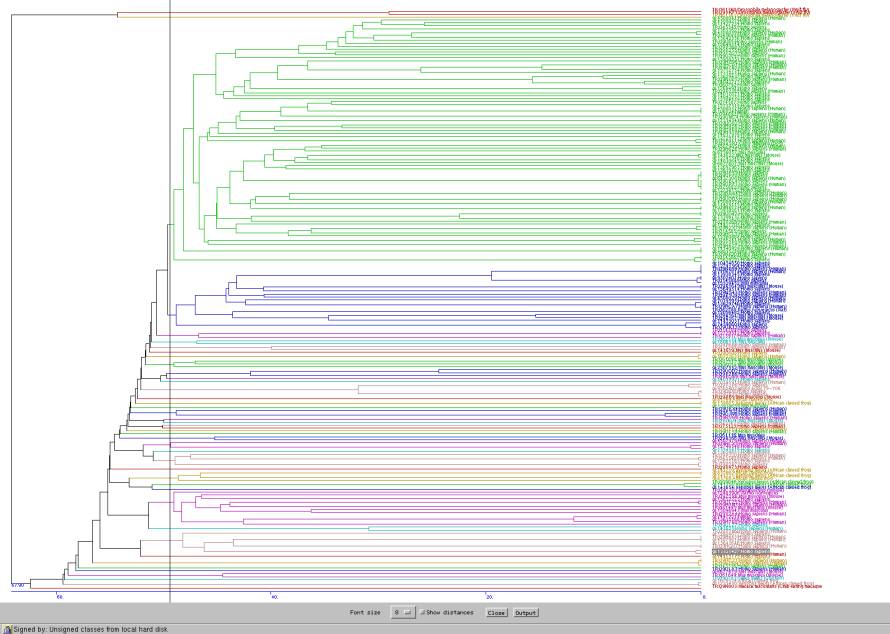
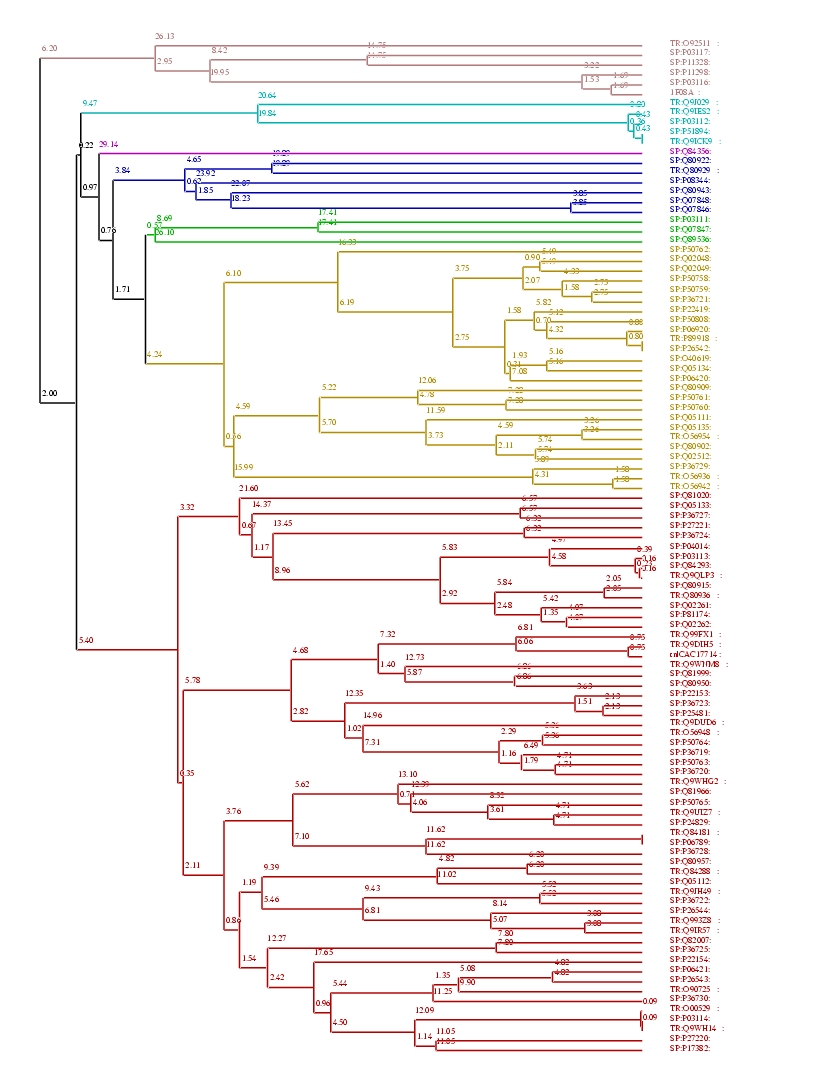
Fig 2. The homology tree of zinc finger DNA-binding proteins super family. There are no significant separate clusters in this super family. All proteins have approximately 50% sequence identity with each other. This suggests that the proteins are structurally have the same fold but might serve many different functions along with binding with DNA.
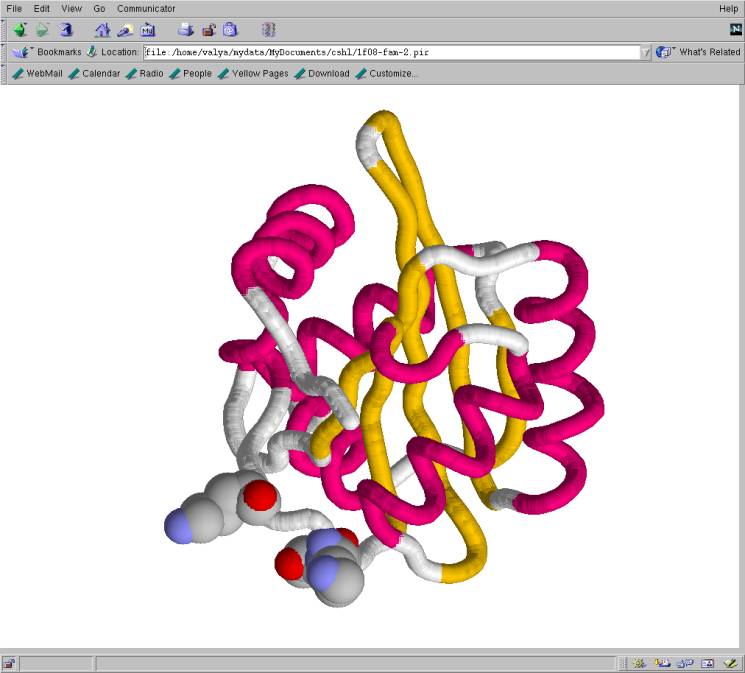
Fig. DNA binding domain of the replication
initiation protein E1 from papillomavirus (Enemark et. al, 2000).
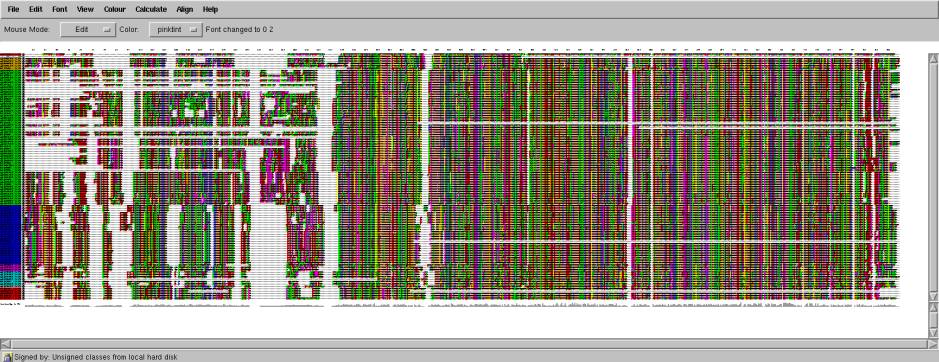
Fig .104 proteins of super family of the Replication Initiation Protein E1. Each residue colored by residue type. The proteins grouped by sequence identity in several families and each family colored by separate color (left column). It is possible to identify 6 different families, two of them are large containing 57 proteins (green) and 25 (blue) and the other contains 4-6 proteins.
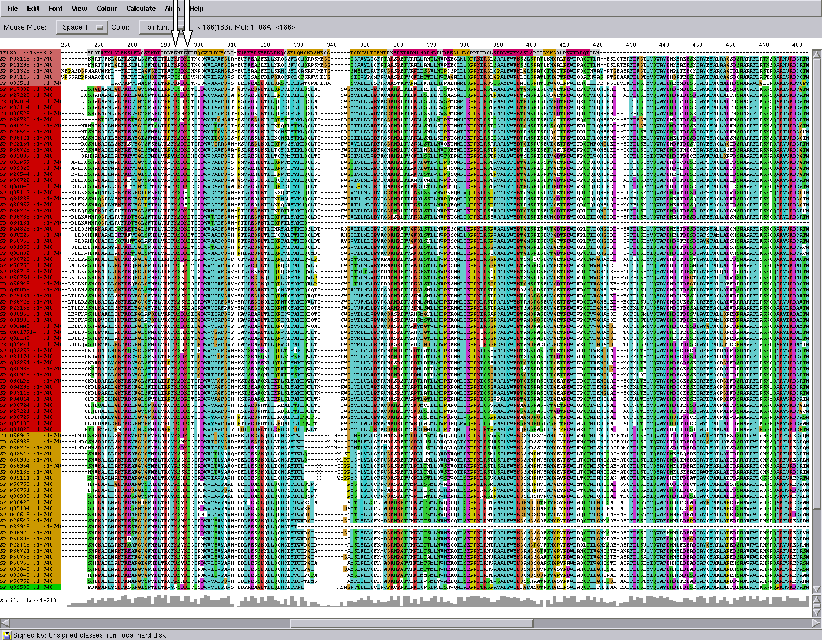
Fig. Part of the multiple alignment of the replication initiation protein E1(E2) super family corresponding to the crystal structure of 1f08. First sequence is 1f08 protein, its secondary structure is colored, red is alpha-helixes and yellow is beta sheets. White arrows point to residues K183, K186, and T187 interacting with DNA.
------------------------------------------------------------------------------------------------------------
Valentin A.ILYIN, Ursula Pieper, Ashley C. Stuart, Marc A. Martí-Renom and Andrej SALI. Visualization and Analysis of Multiple Protein Structures and
Sequences by ModView. 2001 (submitted).
Kim, C. A., Berg, J. M.: A 2.2 A resolution crystal structure of a designed zinc finger protein bound to DNA. Nat Struct Biol 3 pp. 940 (1996)
Enemark, E. J., Chen, G., Vaughn, D. E., Stenlund, A., Joshua-Tor, L.: Crystal Structure of the DNA Binding Domain of the Replication Initiation Protein E1 from Papillomavirus Mol. Cell 6 pp. 149 (2000)